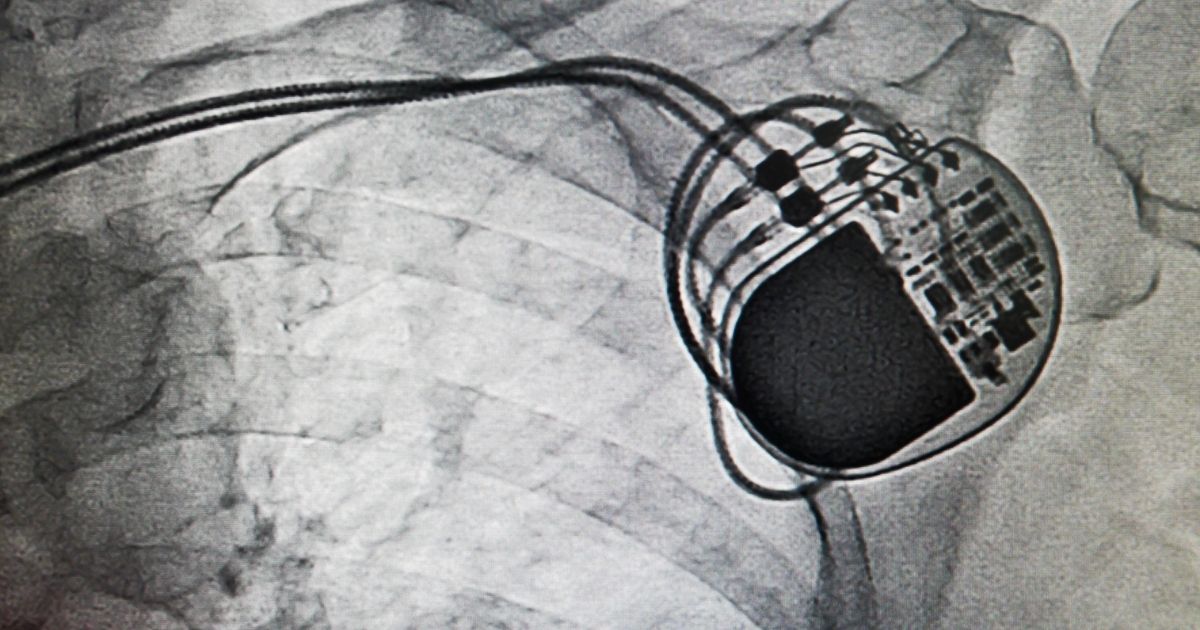
The HeartWare ventricular assist device (HVAD) is a mechanical pump that is surgically implanted in patients who are suffering from a weak or failing heart. The device was approved by the Food and Drug Administration (FDA) in 2012 and has been implanted into more than 19,000 patients. However, after discovering serious problems with the life-sustaining pump, the FDA sent a warning letter to the manufacturer, which stated that they would alert other federal health agencies about the problems. No such alert was released. As a result, government agencies continued to implant the device in new patients, even though there was clear evidence of serious problems, including injuries and fatalities.
The HeartWare HVAD pump is surgically implanted inside the chest and connected to the heart. The pump uses centrifugal force to pull blood from the left ventricle and send it to the aorta. The pump is run by an external computer called a controller, which is connected by a small cable that passes through the skin on the upper abdomen. Once the device is surgically implanted, it takes over the job of pumping blood from the left chamber of the heart to the rest of the body. The HeartWave pump is smaller and lighter than other ventricular assist devices, which makes it possible to implant the device directly next to the heart rather than in the abdomen. This reduces the risk of infection. Most patients who receive a HeartWave HVAD are able to return to school or work since it is an implantable device.
Medtronic, the company that manufactured the HeartWare HVAD, pulled the pump system from the market last year after a number of Class I recalls and reports of patient injuries and fatalities associated with the device. The Class I recalls resulted in reports of 91 injuries and 15 deaths related to the defective medical product.
In addition to the ongoing device failures, there has been a higher incidence of neurological adverse events and mortality with the HVAD system compared to other circulatory support devices. The FDA urged providers to stop using the HVAD pump after Medtronic received over 100 complaints about the device, including a delay or a failure to restart the pump. The agency advised health care providers to use the Abbott device instead of the HeartWare HVAD.
Medtronic had previously stated that even after the warning letter was issued, the company believed that the benefits of the HVAD still outweighed the risk to patients who were suffering from severe heart failure. After considering the findings and the availability of safe and effective alternative devices, Medtronic agreed to stop the distribution and sale of the device.
In addition, the company is working with Abbott Laboratories to make sure that patients who need the pumps have access to the HeartMate 3. The company is also developing a program that will provide financial assistance for eligible patients.
In the Medtronic letter, the FDA instructed health care providers to stop all new implants using the HeartWave HVAD and use an alternative device instead, like the Abbott HeartMate 3 device. It also advised patients who already have the HeartWare device implanted to continue to use the device components and contact the company for controllers, batteries, carrying case, and other replacement items.
Medtronic also instructs patients to never disconnect the HVAD pump from two power sources at the same time and to have a back-up controller available and fully charged with spare batteries available if needed. It is not recommended that patients have the device explanted, although decisions may be made on a case-by-case basis after considering the patient’s clinical condition and surgical risks. Unfortunately, the surgery required to remove the device is generally considered more dangerous.
For the estimated 2,000 Americans who rely on the HeartWare device to keep their hearts pumping, there are a range of side effects and health complications that they may experience. The following are examples of issues that patients have reported:
If you are experiencing any discomfort, injuries, or health complications from the HeartWare HVAD pump, you may be eligible for financial compensation by filing a medical products liability claim. There are three types of negligence claims that can be made against the manufacturers of a defective product, including a manufacturing mistake, a dangerous product design, and a failure to provide adequate warning about possible risks associated with the device.
In addition to the fact that the HeartWare HVAD pump may have a significant design flaw, which led to numerous injuries and fatalities, the company failed to warn patients and health care providers of the potential risks after the FDA sent a warning letter. This puts thousands of patients at risk. In order to have a successful claim, you must be able to prove that you suffered injuries, that the medical device was defective, and that the defect in the device caused your injuries. To do this, it is recommended that you take the following steps:
Having a skilled and experienced lawyer on your side means that you will have the best possible chance of having a successful settlement outcome. The following are some of the ways that an experienced lawyer can help:
The costs associated with a defective medical device can accumulate very quickly, particularly if you are hospitalized and are unable to return to work for an extended period of time. Knowing that your injuries could have been prevented if the defective medical device was taken off the market before it was implanted makes these losses that much more devastating. A successful claim will ensure that you are compensated for the following:
If you have health complications from a ventricular assist device, do not hesitate to contact our Philadelphia defective medical device lawyers at Brookman, Rosenberg, Brown & Sandler at your earliest convenience. To schedule a free consultation, call us today at 215-569-4000 or contact us online. Located in Philadelphia, we serve clients throughout New Jersey and Pennsylvania, including Delaware County, Chester County, and Philadelphia County.